
Protea Neriifolia var. Pink Mink Photo by the Author
Upon Natural History
 Harry Levin Florida Wildflowers presents Dr. Harry Levin's essay "Afterthoughts and Conjectures upon Natural History." This is one of 10 ground -breaking essays in natural history published for the first time. These essays are accessible from the contents page, along with his philosophy of how an engineer can analyze, synthesize, and contribute to new scholarship in the natural sciences, Michael E. Abrams |
By Harry Levin Essay Number 10 Copyrighted Prologue The Cretaceous is the “bank and shoal of time,” upon which these essays end. Yet for the insatiable reader (if such be he or she), this essayist seeks further to reexamine the past. To explore late-Paleozoic and Mesozoic lore, I bring afterthoughts and conjectures to the fore, and proffer ‘sorted morsels more, anent obscure occurrences of yore. Thus, the first person singular enters here. “This rover crossed over” to the sunny side of the street. Continents Adrift I have read that it was axiomatic until 1970 that continents did not move. Yet Macbeth (Macbeth III iv) did disagree quite early: “Stones have been known to move . . . ” Even more, I enjoy quoting Venkata Rao (1) with all his pertinent implications, to the effect that Sir Joseph Hooker in 1860 believed that the existence of similar flora on widely separate lands could not be explained without supposing that they once formed a continuous expanse. Then too, in the early 20th Century, Alexander Du Toit (2) used his travels and his pen to jar loose the continents. The above tempt me to cite Antonio Snider-Pelligrini, who, perhaps earliest, in 1858, wrote “La Creation et Ses Mysteres Devoiles.” He dared place Australia 300 million years ago on the east coast of Africa. The Snider-Pelligrini reconstructions were based on contour fit and on fossil similarity in coal deposits. I am indebted to Continental Drift (3) for this interesting citing of another unsung forerunner to Alfred Wegener. |
1
Reproductive advantage, upon its establishment in nature, can rank as the most powerful stratagem for ascendancy of a phylum, a class, an order, a family, a genus, and a species. It often starts with a mutant species. I pointed first to a species of fish, a species of the genus Etroplus. It adapted to breed in brackish waters, a great advantage indeed, for it could safely reproduce beyond the reach of saltwater predators. Some species of its close relative, the genus Paretroplus, even outdid it; for they found that they could further adapt to breed in waters almost entirely free of brine, thus opening a real opportunity for ascendancy of their family of fish, the Cichlidae, to spread across Africa and South America in vast numbers (4).
I leave it to the reader to fill in examples along the way; and I leap forward to the subclass or superorder, the dinosaur. Herds of herbivores and their herding theropods swam north across the Western Tethys onto Laurasian shores to nest their eggs and juveniles, safely away from therapsid and mammal predators that could not follow over water. Then, in season, the herds went south again to Gondwanaland to feed. Thus by reproductive ploy, by the newly found sheer numbers of their theropods, they overwhelmed the therapsids.
But most far-reaching of all is reproductive innovation at the phylum level. By virtue of evolving the bisexual flower – Hermes and Aphrodite enclosed in each and every such flower – the angiosperm became dominant upon the earth by reason of new independence: enhanced ability to diversify and greater freedom to radiate rapidly far and wide. It started with a mutant species.
2
A noteworthy biogeographical situation involves Sumatra and Borneo and the plant family Nepenthaceae. It is the biotic disjunction known as the “Wallace Line.” Sumatra and Borneo, among the Sunda Islands, are neighbors, separated by roughly 500 km of Indian Ocean. The Sundas include Sumatra, Java, Borneo, Sulawesi, Bali, Lombok, and smaller islands.
Sumatra and Borneo present a puzzle of fauna and flora of two different geophysical derivation. The biota on Sumatra radiated from a gigantic landmass, Austosunda (Essay # 2) that attached to Laurasia about 150 million years ago. The biota on Borneo radiated from the Indian Plate about 85 million years ago. Thus, a phytogeographic disjunction (bimodal distribution pattern) was created by the separate debarkations of the above two landmasses -- 65 million years apart – upon the shore of Laurasia. The presently termed “Australasian” and “Cathaysian” flora and fauna denote this disjunctive phytogeography on Sumatra and Borneo, respectively. The adjacent presences have often been delineated with no little wonder ever since noted by Alfred Russell Wallace and has been termed a “Wallace Line” phenomenon.
Currently there are more than 130 species of Nepenthes, about 100 of which are scientifically accepted. Of these, more than anywhere else, 11 are endemic only to Borneo and 6 are endemic only to Sumatra. The Wallace Line appears to prevail.
Austosunda brought a store of Nepenthes to Laurasia and eventually to Sumatra. It had biologically rifted from Gondwanaland about 280 million year ago, as told in essay. Hence, the antecedents of the Nepenthes on Sumatra originated more than 280 Mya.
In distinction, these Sumatra Nepenthes are older sisters to the Borneo Nepenthes. The antecedents of the Borneo Nepenthes had voyaged on the Indian Plate, which rifted later than Austosunda from Gondwanaland and arrived 65 million years later upon the shores of Laurasia.
3
In conjecture, was Costa Rica once a part of South America? Did it break off from the west coast of South America between Chile and Peru? Did it then drift northward to become an integral part of Central America?
To these questions, I can offer affirmative thought, not answers, by bringing concurrent biogeographical events into focus. They are worth recounting here briefly to encourage further research.
Before the rise of the Andes that began less than 65 million years ago, Peru and Chile, were wetlands and deltas, a moist host to many genera of the angiosperm families Hydnoraceae (5) and Balanophoraceae (6), which are root parasites. Today both families have genera that are endemic to both Costa Rica and South America. In conjecture, both parasitic families in the ancient past rooted westward across South America, and onto a “Costa Rica block,” then contiguous with the western coast where now are Peru and Chile. The Costa Rica block then drifted away to become part of Central America.
I identify the Costa Rica block with the “Chorotega block,” a large portion of Costa Rica. It has an oceanic-crust basement that sets it apart geologically in Central America from the blocks to which it adjoins. These other blocks, such as the Chortis block at its north, have continental-crust basements. The difference in basement composition thus reveals an allochthonous nature of Central America. The region also is suffused with silicic volcanism.
A Hydnoraceae species Prosopanche americana is native to Costa Rica in the Braulio National Park at 500 m elevation. That same species is also native to Argentina and as far north as Bolivia and Peru. Three genera of Balanophoraceae too are native to South America and Costa Rica, They are Corynaea, Helosis, and Langsdorffia. (Ancient ancestor to all three may be the Amborellaceae, extant but only in New Caledonia.)
Both the Hydnoraceae and the Balanophoraceae join in telling that Costa Rica was once attached to Western South America.
4
Vastly more than mere conjecture, the Balanophoraceae bring forth the reality that, in effect, their very presence in the Permian and in the Triassic stubbornly and undeniably negate the doctrine of Cretaceous origin of the angiosperm. It is in Figure 1, as authored by Bruce Cornet, paleobotanist and palynologist (7), that the Balanophoraceae flout Cretaceous origin.
In Figures 2, the remarkable root parasite family, Balanophoraceae, of order Balanophorales, is charted. Today they are present in India, Southeast China, Indochina, New Zealand, New Caledonia, Madagascar, Africa, South America, Central America, and the West Indies, as well as, as noted above, Costa Rica. The world-wide distribution of the Balanophoraceae strongly indicates that 280 million years ago and earlier, the family had propagated over Gondwanaland from one end to the other, as had many other ancient angiosperms alive today. Figure 3 is an artist’s rendition of a genus of Balanophoraceae.
|
Figure 1
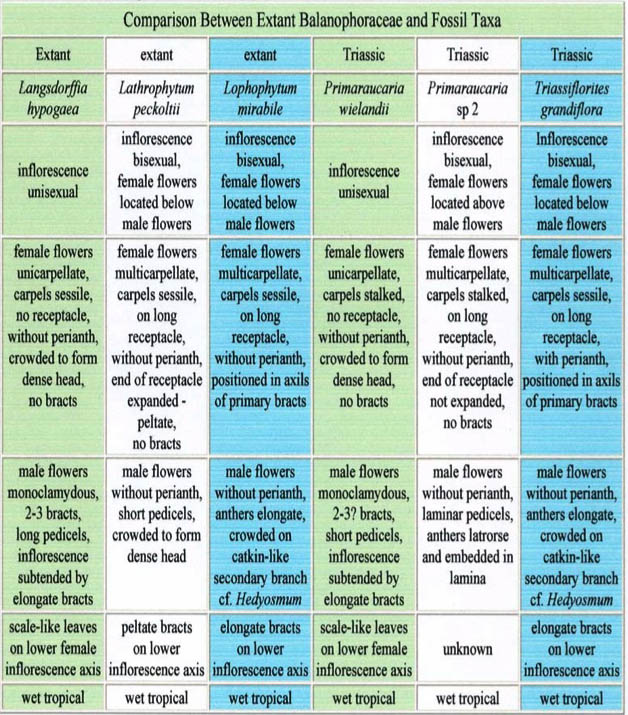 |
5
| Figure 2 Distribution Map Balanophoraceae 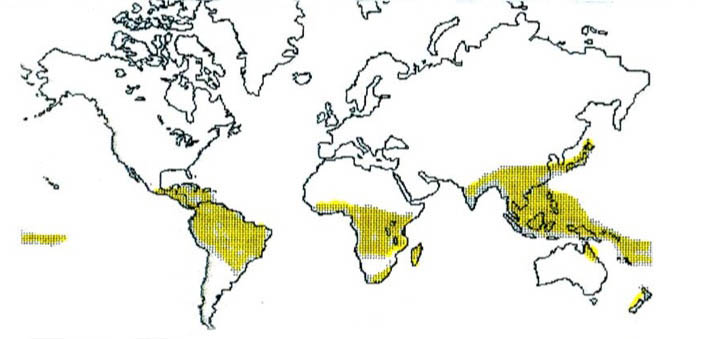 |
Figure Three |
Balanophoraceae Hachettea austrocaledonia, endemic only to New Caledonia. Chromolithography by Auguste Faguet, Paris, 1886. |
6
The earliest angiosperm families shed light on the long-unresolved question of monophyletic (single-origin) or polyphyletic (multiple-origin) evolution of the angiosperms. The evidence here points to monophylesis. Ronald Melville (9), author of the gonophyll, or “sex leaf,” theory of angiosperm origin, in his quest for an ancestral angiosperm, acknowledged the families Magnoliaceae and the Ranunculaceae as examples of stark divergence. But these two families are comparatively newcomers upon the scene. Although they are ancient, indeed, by almost all standards, they are, nevertheless, advanced families. Melville should have gone much farther back – more than 100 million years farther back – to two families within the order Magnoliales.
These families are the Winteraceae and Degeneriaceae, two of the most ancient families for which there is present-day record, a record that is read in their living flowers and not in their fossils. Here exist two significant angiosperms that attest to convergence, to monophylesis. I ascribe to the Winteraceae and the Degeneriaceae antecedents the same approximate date of origin – namely, the late Devonian more than 360 million years ago.
Primitive, indeed, the Winteraceae have no vessels (water-carrying tubes) in their woody (xylem) tissue. The embryos of Degeneriaceae have three or four cotyledons, a most unusual feature. The Degeneriaceae too have a mark of earliest primitive nature in their leaf-like three-veined stamens and carpels. Indeed, among both Winteraceae and Degeneriaceae genera, in the gynoecium, there are rows of ovules on each side of the midveins of their leaf-like carpels; and the carpels are folded along the central axis, with the seams facing the center of the flower (a conduplicate carpel).
These forms are reminiscent of Melville’s “pre-angiosperm transformations” of leaves and bracts into androecium and gynoecium (in his theory of the origination of the flowering plant from a seed fern precursor). Melville’s gonophyll, or “sex leaf,” theory is that the flowers were first built of modified seed fern leaves bearing either ovules or microsporangia.
Furthermore, the seeds both of the Winteraceae and the Degeneriaceae contain a minute, relatively undifferentiated embryo, which occupies only a small part of the seed at maturity.
Here again, recall the family Proteaceae in earlier essays. The Winteraceae have four pollen sacs, suggestive of the four anthers (pollen sacs) adnate to the perianth walls of the Proteaceae. And unisexuality (a throwback to the seed fern) occurs in a genus of Winteraceae, as it does in several genera of Proteaceae.
7
The Proteaceae have been described as taxonomically odd and isolated, but they belong among the early angiosperm families. I surmise that all three families above are late Devonian, the important antecedents of which are lost to earth history.
Like the Proteaceae, the other two families are alive today. They are both of order Magnoliales. The Winteraceae, have 7 or 8 genera and 88 to 120 species, all but four of which are native to Southeast Asia and Australasia. The Degeneriaceae, have only two genera, both large trees found on Fiji. Both families are kindred to the Magnoliaceae.
In summary, the Ronald Melville gonophyll theory of the origin of the angiosperm appears to be sustainable both in its critical biogeological time requirement and in its early examples. The Proteaceae, Winteraceae, and Degeneriaceae are among the earliest known angiosperms. They are today essentially indigenous to the southern hemisphere, just as they were at the time of their origins.
The biogeophysical milepost of the time of origin of the angiosperm is the adjustment needed to re-read the earth’s clock for the true accounting of the interactive histories of flowers, mammals, dinosaurs and birds. Thus, the remarkable presence of the angiosperms over late-Paleozoic and Mesozoic history calls for reexamination of man’s records of earth history in order, for the first time, to take into account the profound effects of the angiosperm on the evolution of mammals and dinosaurs. It is impossible to understand even the dinosaur and bird relationship without recognizing the influence of the angiosperm.
8
___________________________________________________________________________________________
The Dinosaur-like Nesting Habits of the Largest Ratites
I call upon the largest ratites, the ostrich, the emu, and the cassowary, to tell of the nesting habits of dinosaurs– to display the breeding of dinosaurs by nesting features that these birds share in common. The ostrich has been assigned to the order Struthioniformes; the emu and the cassowary, generally to the order Casuariformes. The ostrich is African; the emu and the cassowary are Australian, the latter also a native of New Guinea. Nominally their mature weights run from 70 to 120 kg., from cassowary to ostrich.
For birds in general, characteristic dinosaur nesting patterns persist. Among these are two-parent, long-time incubation of eggs; sheltering of hatchlings; and regurgitate feeding. The nesting patterns of today’s largest ratites, ostrich, emu, and cassowary, reflect with amazing consistency the nesting history of the dinosaurs, especially the vulnerable long brooding times essentially at ground level.
Like dinosaurs, the above ratites all travel in herds and colonize tightly during the breeding season. Emus and cassowaries are excellent swimmers. These ratites all nested on the ground. For nesting, the cassowary prefers rainforests and swamps; ostriches and emus are inclined to seek grass. The clutches of the cassowary may contain 4 to 8 eggs; the emu, as many as 25. On the other hand, ostrich clutches may be communal, with as many as 60 eggs. By chicken standards, the eggs are of huge size.
The clutches of the ostrich and the emu are bare and shallow, often shielded from predators by grasses and weeds. The cassowary clutch may be a slightly raised platform built of plant debris. Incubation of ostrich, emu, and cassowary eggs takes about 42, 55, and 65 days, respectively. The males have important roles, even predominant, in the incubation. Both parents and other herd members share in feeding and protecting the hatchlings in the nesting area for periods even as long as nine months. Nest predation by cats, dogs, foxes, feral pigs, reptiles, etc. is a serious concern and requires constant vigilance. (Avoidance of nest predation was crucial to the dinosaur in its prolonged nesting on the ground.)
9
__________________________________________________________________________________________
Retrogressive Behavior and Crestal Atavism in Birds
There had not been a quantum jump backwards in evolution among birds. Nature offers numerous living examples of gradual steps backward to diminished flight efficiency, to “short-hopping,” and eventually to “grounding.” These examples include quail, turkey, partridge, peafowl, chickens, etc. – birds that have opted to reduce air travel time in their quest for survival.
Here and there among birds are indications of atavistic morphology, again throw-backs to the dinosaur. The occipital crest is a notable example. I first mentioned crests as a skull feature of ceratosaurian theropods living at the Mount Kirkpatrick site in Antarctica 200 million years ago. Later biped dinosaurs, hadrosaurs, which were abundant in Laurasia, were as tall as 10 meters and strangely crested.
Occipital crests occur today here-and-there among species of diverse birds such as eagles, hawks, herons, ducks, cassowaries, and even penguins. Crests (“casques that did affright the air”) might offer protection in scampering through underbrush, or, in the case of the penguin, in swimming among seaweeds. I am reminded of atavism among flora: for example, the reversion of scattered genera among angiosperms to their unisexual seed fern ancestry of 360 million years ago.
The Oystercatcher and the Frumious Therizinosaur
My thoughts return to the amazing metamorphosis of dinosaur to bird. Some of its aspects are tenuously retrievable, conjectural, yet persuasive. I am fascinated by the bird order Charadriiformes, 16 families of marine and freshwater birds, web-footed, ancestrally feathered black and white – and like dinosaurs in general, nomadic, gregarious, breeding in colonies. Specifically, my thoughts turn to its family the Alcidae, more than 60 million years ago offspring of coelurosaurian theropods – perhaps the first flyers.
Yet there is another family among the Charadriiformes that excites my curiosity. It is the family Haematipotidae, the oystercatchers. These birds, by their very name, patrol the tideline zone of the coastal littoral and dig up, open, and partake of clams, cockles, limpets, mussels, and oysters by means of their stout, thick, long, and sharp-ended bills (and partake of crustaceans, worms and other invertebrates as well).
I puzzle a while. Where among the dinosaurs might there have been the antecedent for such bird behavior? Then there sprang to mind the coelurosaurian therizinosaur, that frumious bandersnatch, “the claws that catch,” the so-called freak, perhaps maniraptor, that plays hob with phylogenetics.
10
__________________________________________________________________________________________
The therizinosaurians might have used their strong legs, their long arms, their enormously lengthy claws, and the toothless beak fronting their snouts, as carnivores, to dig, pry loose, grasp, open, and feast on seafood while standing in shallow water. And then again, as herbivores, they might have used those lengthy claws and toothless beaks to pull down and snip off branches of small trees and large bushes.
Then, upon surmise, I took a more comprehensive view of the oystercatcher behavior pattern. Here is, namely, the enhancement of aquatic abilities that feathers provide in acquisition of seafood. While, in precept, feathers were developed first for insulation and finally for flight, this intermediate use of feathers should not be overlooked.
In the Cretaceous, as the Gondwanaland continents drifted farther apart, the increasingly efficient use of feathers made it possible for dinosaurs to travel longer distances by water. At the same time, feathers made seafood increasingly accessible to their enhanced aquatic abilities. At Beipiao (where 120 to 100 million years ago dwelt that therizinosaurian) and elsewhere, feathers made it more conducive for dinosaurs to use the seafood content of cold lakes and watery connections for food supply. Dinosaurs, as their feathers evolved, could better stalk, swim, and dive, and even glide, for the abundant fish, bivalves, univalves, gastropods, shrimp, crabs, insects, and other arthropods reported in those fossil digs. Witness both the evolving oystercatcher and the absurd therizinosaur.
Alas for the Paddle-footed Reptile
The plesiosaurs, ichthyosaurs, and mosasaurs (the latter closely ancestral to snakes) deserve some of the credit for the dominance of the earth by the dinosaurs. These paddle-footed reptiles ranged in size to more than 15 meters; and they guarded the Western Tethys effectively during the Mesozoic by preventing crossings of the seaway by therapsids and mammals. However, earth history pays little attention to their abrupt quitting of the earth at the end of the Cretaceous, 65 million years ago. The flooding effects of the asteroid collision no doubt did much to cause their extinction (except for some deep-sea ichthyosaurs).
But by the time of the asteroid collision, these reptiles were already embattled. They were witness to the lowering of sea level and thus to the diminution of seafood resources during the late Cretaceous. Moreover, they were also witness to the drifting apart of the Gondwanaland landmasses. Alas for them, in the late Cretaceous, the opposing Western Tethys shores were moving farther apart. They witnessed the gradual destruction of their nesting sites on shore – ecological niches that had until then provided quiescent shorelines with lapping waters – nesting sites that were, in actuality, the Western Tethys, configured by contact of continental margins for thousands of kilometers and at least 150 million years. The waters that once lapped quiet shores were replaced by pounding surf and other raging conditions unsuited for safe nesting.
11
______________________________________________________________________________________
Some others I know among reptiles that lay their eggs on shore avoided asteroid extinction by burying their eggs and by not nesting their newborn. In re-visiting The Reproductive Advantage for Survival, I recalled the sea turtle and its breeding habits, an apt example from the reptilian order Testudines. I was struck by the following sagacious thought:
Hatchlings that safely run away
May hatch a batch another day.
of Considerable Magnitude
Can I, among others, acclaim the baleen whale, Balaenoptera musculus, the blue whale, as the champion Gargantuan of all times? Does that cetacean really merit the title as the most gigantic of all creatures that ever lived? Does the whale prevail in competition with dinosaurs? Yes and no; it depends on what variable is measured.
In length, from nose to tip of tail, the blue whale loses by plenty. Some blues may tape at more than 30 meters. But a fossil sauropod, Argentinosaurus huinculensis, found in 1997, is estimated to tape at 43 meters. And another sauropod recently uncovered in Patagonia indicates 51 meters. However, if the criterion is ponderosity, the blue whale is the winner. Some female blues weigh in at more than 150 metric tons. The above sauropod A. huinculensis has been assigned a modest 100 tons. And that Patagonian sauropod, newly found, is estimated here to scale in at only 120 tons.
The weight deficit of the dinosaur comes as no surprise me. It is due in part to its porous and hollow bone structure. Light bones were characteristic of all dinosaurs. Hollow bones contributed in high degree to body air storage and buoyancy. The empty spaces were needed to lighten the huge bulks so that dinosaurs could feed on land and travel considerable distances over land and water to breed. Hollow bones contributed even further to dinosaur adaptability to feeding. For example, the extremely long necks of sauropods were, of necessity, structured light in weight in order to feed high up in trees. Sauroposeidon had cervical bones more than 3 meters long in a 12-meterlong neck. Such sauropod bones are replete with tiny air cells.
The 51-meter Patagonian dinosaur, above, could comfortably remain on all fours while eating fruit, flowers, and leaves at an estimated 18 meters up a tree. Tall sauropods evolved ever taller to answer the challenge of bountiful flower harvests near the tops of extremely tall angiosperm trees, which are here inferred to have reached 18 meters or more in height in Patagonia. And gigantic meat-eaters like Gigantosaurus grew apace. Huge predator and huge prey both abounded in Argentina. The time of this huge dinosaur growth was toward the end of the early Cretaceous, about 110 to100 Mya, during the halcyon days of crossing the western Tethys, north to feed and south to breed.
12
________________________________________________________________________________________
Genetic-Magnetic Interaction in Fauna and Flora
Cattle and deer have been statistically shown to point their bodies in a north-south direction to graze and to rest (for reasons described in Essay #9). The north-south orientations of cattle and deer, when taken together with the north-south migrations of dinosaurs and birds (as described in Essay #5), are manifest evidence that magnetic fields interact with DNA. At the time of dinosaurs, the earth’s magnetic field was significantly stronger than it is today.
Furthermore, I conjecture that the DNA of the dove can identify and remember the lines of force that emanate from the magnetic pole and pass through the dove’s nesting site; and the dove, after distant displacement, can seek out and guide on these particular lines of force to return to its nesting site.
It is highly probable, moreover, that the earth’s magnetic field is somehow involved in the observed homing behaviors of salmon, newts, turtles, and alligators.
In flora, migration (propagation) results from stronger causes than the gentle persuasion of a magnetic field. My thoughts recall the example of a flowering plant, the sunflower Helianthus. Its DNA components interact with the electromagnetic field of solar radiation. Each flower, when in bud, torques its stem (twists its neck, so to speak) as it follows the sun on its diurnal trek across the heavens in rain or in shine. In the words of Thomas Moore:
“The sunflower turns on her god as he sets
The same look that she turned as he rose."
Thoughts on the Miracle of Songbird Migration
'
I attend with pleasure the chirping of thrushes as they fly by in season. Songbirds, in migration, have been noted to fly extremely long distances at great speed overnight. Studies have cited that the purple martin (Progne subis), the wood thrush (Hylocichla mustelina), Swainson’s thrush (Catharus ustulatus), and the grey-cheeked thrush (Catharus minimus) travel more than 300 miles in sustained flight 11 to 14 hours per night over many consecutive nights (10) (11).
For example, the purple martin was measured by geolocators (10) to have flown about 1500 miles southward in five days. (It is interesting that birds have been noted to fly 2 to 6 times faster heading north in the spring as they do heading south in the fall, perhaps because the youngest members of the flock exact a slower pace in heading south.) Bird flights, in general, are northward in the spring to their breeding grounds and then southward in the fall to their wintering grounds.
I contend that the seasonal migration guidance system of songbirds, as well as of other birds, is innate. It is the heirloom of dinosaur DNA awareness of the earth magnetic field begun since early Triassic, about 225 million years ago. From that time on until demise, by natural selection, dinosaur herds crossed over a narrow water channel from Gondwanaland into Eurasia to protective breeding grounds -- in season, north to breed, and again south to feed more abundantly (in Gondwanaland).
Hence, in the deep past, was born the genetic-magnetic migration memory of birds. The dinosaur herd was father to the flock of today’s purple martin. Today’s 40-gram Swainson’s thrush is a distant cousin of the humongous theropod, Tyrannosaurus Rex. Furthermore, the very ability of songbirds to fly is a crucial hand-me-down from their bipedal theropod dinosaur ancestors: namely, their hollow, thin-walled, internally strutted light-weight vertebrae.
The earth’s magnetic field does even more at present to assure the correct orientation for nighttime migratory flight for songbirds (thrushes of genus Catharsus, for example), and other birds smaller and larger. The birds calibrate their innate magnetic compasses for overnight flight at the time of the setting of the sun (12). Night migration may have become prevalent among songbirds over time by natural selection for avoiding predators such as falcons, hawks, and eagles.
In conjecture, I am persuaded by experimental and theoretical studies of others (11) (13) that the above songbirds will lose roughly 30 to 50 % of their body weight as energy expenditure during a nighttime 11-hour, 300-mile flight. In order to enable the next night’s flight, that weight (lost as fat, equivalent to energy) needs to be regained, essentially by restoring the loss of body fat. The lost weight can be recovered only by feeding during the intervening non-travel hours.
Obstacles seem formidable for regaining 30 to 50% of body weight as fat for each successive night of flight by feeding and alimentary tract processing:
• Availability of nutrients can vary greatly in time, place, and weather.
• The energy required for food-seeking itself can be high and variable, depending sensitively on abundance and accessibility.
• Food sources, excluding water, provide variable fat contents of generally less than 35% fat. The balance is carbohydrates, proteins, and non-nutrient materials. Carbohydrates and proteins per se, on combustion, supply only about half as much energy (calories) per gram as do fats. Indeed, carbohydrates in body storage (as glycogen) retain water and so provide much less energy per gram than fat.
From the above, it is evident that fat plays a major role in the migratory behavior of songbirds. Fat is anhydrous and leaves no metabolic waste in conversion to energy. The rapid daily restoration of body fat is a necessity for successive nightly flights. It would, therefore, seem essential, preparatory to each night’s long-distance flight, that ingested carbohydrates and proteins be converted, at extremely high efficiency, by enzymatic processes, mainly to unsaturated fatty acid triglycerides for fat storage. Apparently, for the migration period, songbirds have provided themselves genetically with greatly enhanced metabolic adaptations for fat processing: appetency, digestion, lipogenesis, storage, and utilization.
All the more phenomenal! These songbird migrations are remarkable evolutionary accomplishments.
Asteroid Disaster
The epochal asteroid catastrophe of 65 million years ago, to whatever extent that it was attended by volcanism (14), wrought profound mass extinction. Yet – this afterthought is strictly a conjecture – descriptions of the disaster seem to overlook what, in my estimation, may have been a most important factor in the wide-spread devastation that resulted, namely: the sudden gutting of the “oil tanker” Earth, wherein oil was made and stored in great quantity during the relative mild Mesozoic. There must have been an enormous release of shallow-bedded crude oil to the earth atmosphere and to its surfaces of land and water. It could be a main reason for the extinction of the dinosaur and other aquatic creatures that bred upon wetland shores.
The extreme degree of deadly disaster to the dinosaurs indicates that the asteroid collision occurred at the time when the dinosaurs were most vulnerable – at the busiest part of the long breeding season, when the eggs had all been hatched and the hatchlings were fully dependent. The oil sludge may have quickly coated the hatchling dinosaur bodies inside and out. Today, the rehearsals of lethal effects of crude oil spills on littoral ocean fauna remind me that crude oil could well have been a big factor in that great disaster.
To continue this conjecture, I have found no mention of long-term consequences, of untold animal cancers, resulting from this sudden release to the atmosphere of hydrocarbon-based carcinogens – the release of finely-dispersed oil and sludge particles from the reservoir basins and from the long “gutter” of the Western Tethys; and also release from other shallow waters.
But then I’m visited by a contrary thought: Can any good come out of catastrophe? Could there have been a long-term benefit? By that accelerated natural selection process over an indefinite period of the past, could human beings today have evolved to be more respiratory-cancer resistant? Otherwise, by now mankind might hardly have survived automobile petroleum fumes as he travels the precarious roads to outmaneuver evolution that may lead to a self-destruction which he cannot foretell.
13
______________________________________________________________________________________________________
Counsel
In pleading phyletic gradualism, Charles Darwin wrote (15) “No complex instinct can possibly be produced through natural selection, except by the slow and gradual accumulation of numerous slight yet profitable variations.” Evolution expresses, indeed, the essential totality of ordinary variation; and in nature it is almost never abrupt or “punctuated,” even at times of rapid environmental change leading to intensified natural selection.
I suspect that there may never have occurred in nature an abrupt appearance such as, for example, a polyphyletic Cretaceous origin of flowering plants, except as reversion to some ancestral type. Indeed, mental resort to punctuated evolution may in many cases be an impatient exegesis to fill a gap in knowledge.
Current global changes – geophysical and climatological – emphasize the need for further knowledge and understanding of past events of origin and extinction which have occurred here upon the earth. I hope that all students of earth science and of earth history will, therefore, feel invigorated to search for an earth that was nearer to reality.
“Speak to the earth and it shall teach thee.” (Job 12:18). Much scholarly inquiry waits to be done among grasses, herbs, shrubs, and trees to learn of their evolutionary adaptations to changing geology, climate and competition. And among fauna, penguins, for example, the respiratory and heat exchange engineering of which exceeds human endeavor.
“If we lose our capacity to be wrong, we are not doing the business of science.” Charles L. Drake, late Professor Emeritus, Dartmouth.
14
References
1. Rao, C. V., Botanical Monograph No. 6, Proteaceae, Council of Scientific
and Industrial Research, New Delhi, 1971
2. Du Toit, A. L., Our Wandering Continents, Oliver and Boyd, Edinburgh, 1937.
3. Tarling, D. H. and Tarling, M. P., Continental Drift, A Study of the
Earth’s Moving Surface, G. Bell and Sons, London, 1971.
4. Stiassny, M. L. J., and Meyer, A., Scientific American, 282, 64-69, (1999).
5. Musselman, L. J. and Visser, J. H., Aliso, 12, 317-326 (1989).
6. Nickrent, D. L. et al, J. Am. Botany, 89, 1809-1817, (2002).
7. Cornet, B., “Fossil evidence for root parasites (resembling Balanophoracea)
in late Triassic,” 29 pages, on web site Bruce Cornet, updated March 2007.
8. Vasanthy, G., Cornet, B., and Pocock, S.A.J., Evolution of proangiosperms
during Late Triassic, Geophytology 33 (1 and 2) 99- 113 (2224)
9. Melville, R., Glossipteridae, Angiospermidae, and the evidence of
angiosperms’ origin, Bull. J. Linn. Soc., 86, 279-323, (1983).
10. Stutchbury, B. J. M., et al., Tracking long-distance songbird migration by
using geolocators, Science 323, 896 (2009)
11. Johnson, C., Scientific analysis of the efficiency of bird flight, Internet:
Mb-soft.com/public 3/birdeff.html, updated 12/25/08
12. Cochran, W. W., Mouritsen, H., and Wikelsky, M., Science 304, 405-408,
(2004)
13. Salt, G. W. and Zeuthen, E., The respiratory system, Chapter 10 in text:
14. Officer, C. B. and Drake, C. L., Terminal Crustaceous environmental events,
Science, 227, 1161-1167, (1985).
15. Darwin, C., The Origin of Species: By Means of Natural Selection,
Chapter VIII, Random House and others