By Harry Levin
Copyrighted
Prologue
The Proteaceae have a timeless beauty that brings to mind hillsides in
cool sunshine flowered with red, yellow, and white ancestors of Protea
(first frontispiece). They were, in the words of Marie Vogts (1),
“present in Gondwanaland long before it began breaking up 300 million
years ago.”
The unbridled loveliness of Protea ennobles this planet this very day
with
that beauty.

Dr. Harry Levin
|
The Proteaceae are among the most primitive families
of the angiosperm,
the flowering plant phylum of the plant kingdom. Carl Linnaeus, the
father
of taxonomy, is said to have been troubled by the multiformity of
flowers
of the Proteaceae.
Two frontispieces show in contrast the stark reality
that
troubled Linnaeus. They reveal startling differences in the morphology
of inflorescences of genera of Proteaceae.
With age may come wisdom; and the Proteaceae give meaning to their
message: “Earth’s history is written in her flowers.” Proteaceae are
named after the god figure Proteus, and he could change his form.
|
Like
Proteus, the Proteaceae retained the very essence of their nature while
adapting to
harsh environmental challenges. The specialized sex-organ identity of
the Proteaceae has not changed in over 300 million
years.
Befitting the everlasting Proteaceae, Shakespeare, in his wisdom,
wrote: “nothing ‘gainst Time’s scythe can make defense save
breed;” and “Nor shall Death brag thou wand’rest in his shade,”
(Sonnets 12 and 18). As treasures rare, ancient – and endangered –
the Proteaceae urgently
need thoughtful protection from the unrestricted encroachment of man
and his works into their fragile habitats in Australia, Africa, and
elsewhere.
1
|
Frontispiece
Protea
neriifolia variant

There are 136 species of
Protea, all endemic to South and Central Africa.
The flower head
of Protea is an involucred flower cluster.
All
photographs are the
work of Dr.
Levin
and are protected by copyright. Inquiries
about the article and
its photographs
should be made to Dr. Levin whose
email address appears
above.
2
|
Second Frontispiece
Flowers of the Firewheel
Tree
Wheel-spoke flowers of the
Australian firewheel tree, Stenocarpus
sinuatus, at mag. x 4. A style
emerges from an immature flower.
On each style is a superior
positioned (brownish white) ovary.
Inside a "tennis ball" at the top of
each flower nest 4 anthers and
an immature stigma.
At the bases are four black
nectar glands, slightly visible.

The firewheel is
pollintated by
the brush-tongued rainbow lorikeet,
Trichoglossus haematodus, in
reward for nectar.
3
|
Introduction
The plant family Proteaceae represents the history of the angiosperm
phylum simply by telling their own story. Their flowers were one of the
earliest experiments of the angiosperms in bisexual floral structuring.
Alive
today, they have no close relatives. Odd and isolated, they have been
referred
to as “bizarre.”
Current doctrine holds that the Proteaceae, as members of the
angiosperm phylum, originated in the Cretaceous – perhaps because
fossil flower records of the late Paleozoic and the early Mesozoic are
almost
nonexistent. Fortunately, however, an independent historical record is
kept within
the live flowers of the Proteaceae themselves. These flowers carry
within themselves a unique and unmistakable sex organ identity
unchanged over
vast
intervals of time and distance.
In hypothesis, the Proteaceae originated in the late Paleozoic, 360 to
250 million years ago – before the coming of the dinosaurs.
The last
50
million years of that era were marked by the Permo-Carboniferous Ice
Age (also termed the Gondwanaland Ice Age) with its pervasive ice
fields that
covered much of the southern hemisphere. Today the Proteaceae are
distributed
over the southern hemisphere in remarkably strange ways that can be
explained by the impact of that epochal glaciation and the drift of
continents
and smaller continental blocks. The geographic distribution of
Proteaceae genera is inexplicable under the doctrine of Cretaceous
origin.
Botanists have classified the Proteaceae into two subfamilies and
fourteen tribes. The two subfamilies are the Proteoideae and the
Grevilleoideae, referred to here simply as P-oides and G-oides. A third
frontispiece
chart shows the present-day distribution of P-oides and G-oides over
the
world.
For ready distinction, here Proteaceae genera native to Australia are
also named Austrops; those native to Africa, Afrops. For example, a
P-oide
genus native to Australia is an Austrop P-oide; a P-oide genus native
to
Africa is an Afrop P-oide. No Austrop is native to Africa; no Afrop is
native to
Australia.
|
|
Present
World Distribution of Proteaceae
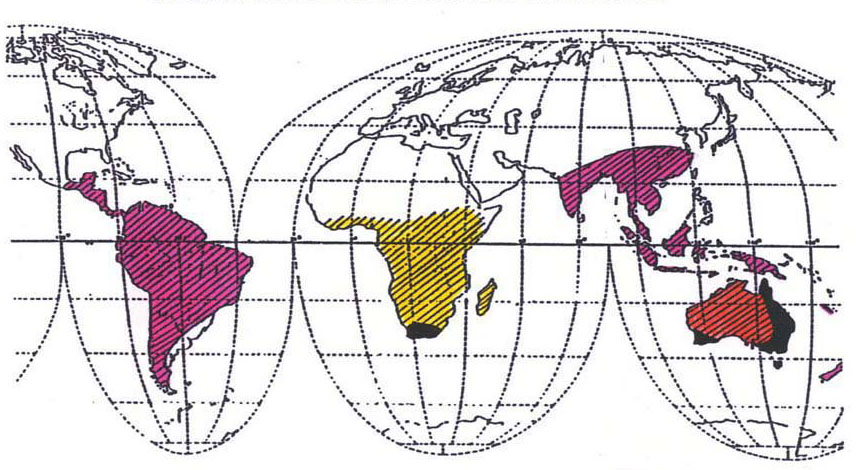
Chart is a
composite of sources
|
P-oides
(Proteoideae) |
|
G-oides
(Grevilleoideae) |
|
P-oides and
G-oides |
|
Regions of
Dense Distribution
|
|
C.
Venkata Rao, Proteaceae p. 3 (1971)
L.A.S. Johnson and B.G. Briggs,
Bot. J. Linn.
Soc. 70 83-182
(1975) |
5
|
With the aid of photography, this essay demonstrates the
remarkable
similarity, hence constancy, of the reproductive structures of the
flowers of the Proteaceae of Australia and Africa. The similar, yet
unique sex
organs of the flowers of Australian and African genera speak of a land
passage
long ago traveled by Proteaceae between Australia and Africa. This
sameness
of floral elements has been preserved to the present time despite the
remarkable chronological separation of several hundreds of millions
of
years and
the interposition of the Indian Ocean.
The leaves of present-day Proteaceae, within genera, display a sequence
of evolutionary curtailment of form and structure in accord with the
premise that the foliate structure of the Proteaceae simplified
defensively in
adapting to the gradual but varied encroachment of the
Permo-Carboniferous Ice
Age of 300 to 250 million years ago. Several leaf
form sequences will be presented by photos in support of the
premise.
Banksia ericifolia
Consider first Banksia ericifolia, an Austrop G-oide shrub or tree,
which may grow to a height of about 3.5 meters. It is commonly known as
the heath banksias, and it is found in large numbers today in Victoria,
Australia.
Banksia ericifolia is chosen because it bears the imprint of the Permo-
Carboniferous Ice Age. It is a survivor of the last glacier foray into
New South Wales (250 million years ago) and is a living example of
how
the icy cold of 300 to 250 million years ago altered the Proteaceae in
reach. B. ericifolia came as close to the glaciers of New South
Wales as
nature’s chemistry would allow. Ice molded it into an entity hard and
frugal.
Its very frugality caused it to husband only the essentials of growth
and reproduction. Among these are the basic flower characteristics that
unite the Austrops of Australia and the Afrops of Africa, while setting
them off
from all other flowering plants.
B. ericifolia’s flowers exemplify “evolutionary simplification”.
Indeed, evolution does not perforce lead to ever increasing complexity.
It does
at times retrench or regress in form or function. Thus it may enable a
species to survive in an increasingly hostile
envionment.
Figure 1 shows a flower spike of B. ericifolia. It emerges upward among
needle-like leaves. It is about 14 cm. tall and looks like a
small corn cob from the distance.
6
|
This
flower spike is at an early stage of reproduction and bears about 1000
flowers. In magnification, Figure 2 shows immature flowers on the
spike. The flowers are in pairs – a feature
that
distinguishes these and other G-oides from P-oides.
A pair of flowers illustrates (in Figure 3) the location of basic
flower parts. There is the perianth, which looks like a musical
quarter note: a thick grayish stem with an angled cup at the upper end.
There is the style, a
bent conduit, a female part of the flower. Characteristically, at this
early
stage of maturation, the style has not yet entirely escaped the
perianth cup. At
the head of the style, a tiny knob, termed a “pollen presenter,”
remains
within the perianth cup. The style of B. ericifolia is like a
wicker,
hard and wiry, forged by the cold.
Are these flowers? Yes, but they are hardly recognized as such
except by the trained eye. The perianth, stark as it is, is really a
flower tube, the
outer part of the flower. And these flowers are bisexual.
Within their
frugal apparatus, hidden and protected from the cold, are
all the sex organs necessary for reproduction of the plant. The male
sex organs, called “the androecium,” consist only of four anthers
joined to the inside
wall of each of four segments that form the angled cup of the perianth.
The anthers apparently are without filaments. They are mature and are
producing
pollen.
The female sex organs, called in sum “the gynoecium,” consist of the
style with a minute ovary at its lower end and the stigma at its upper
end, positioned at the very tip of the pollen presenter. At this
early
stage shown in Figures 2 and 3, the pollen presenter, nestled (unseen)
within the
cup of the perianth, is closely surrounded by the four anthers.
The
stigma itself is not yet receptive to fertilization. The male sexual
organs become
functional before the female.
In the next stage of maturation, the style will “flip out” of the
perianth cup. The flower then will be said to be “laid open”, and the
pollen
presenters will be covered with pollen. Figure 4 in magnification shows
a flower spike
for which the flip-out is under way. The flowers, in a gradual,
orderly
manner, are being laid open from the bottom to the top of the spike.
Here the
pollen presenters are barely visible atop the open styles. They
are minute
among the Proteaceae.
7
|
Figure 1: This is a flower spike of B. ericifolia, emerging
upward among
needle-like leaves. About 14 cm. tall, it resembles a small corn
cob from a distance.

8
|
Figure 2: Magnified, immature flowers on the spike of B.
ericifolia. The flowers
are in pairs, a feature that distinguishes these and other G-oides from
P-oides.

9
|
Figure 3: Banksia ericifolia immature plucked flowers, mag. x 6.5.
Compare with frontispiece.

10
|
|
Figure 4: Banksia ericifolia styles
"flip out" as maturation
advances
from bottom to top of spike.

11
|
Most significantly, the floral
features of the G-oide B.ericifolia are common not only to Austrops but
also to Afrops, with the exception of a few unisexual genera, which are
perhaps atavists of ancient seed ferns. In particular, the
perianth/style relationship is the readily recognized
signature, a hallmark, of both Austrops and Afrops in form and
function. The
austere simplicity of the flower of B.ericifolia (in defense against
the ice)
can best be appreciated by comparison with the more intricate and
elaborate
flowers of selected Afrops and of other Austrops to follow. But first,
the
leaves.
Intimations of an Icy Past among
the Leaves
The genus Banksia attests to the premise that Proteaceae leaves, in
form and structure, reflect various degrees of adaptation to the
glacial
conditions of the Permo-Carboniferous Ice Age. Even today, they range
from the
primal rainforest large, compound leaves to the needle-like leaves of
B.
ericifolia.
Accordingly, a botanical comparator, a “leaf form sequence,” of
Banksiae is presented to illustrate both effect and degree of ice field
encroachment. Banksia ericifolia, Banksia spinulosa, Banksia
integrifolia, and
Banksia burdettii are shown in Figures 1, 5, 6, and 7, respectively, in
sequence of increased leaf elaboration and presumably of increased
distance from
ice field. While this paper does not cover the full range of the change
for Banksia, it does point out that those species that might once have
stood farther away from the ice’s edge have broader and more elaborated
leaves than Banksia ericifolia. Those other Banksiae can be described
as progressively less austere, or more expansive. The premise of
simplification in proximity to ice is supported not only by Banksiae,
but is also
evident, as will be shown, in the Austrop Grevillea, the largest genus
of
Proteaceae.
The term “ericifolia” means heather leaf. The short needle-shaped
leaves of B. ericifolia are like those on the heather of the blustery
moor and on
fir trees and other cold-adapted conifers. The Afrop P-oides show a
parallel example of leaf simplification in Leucadendron ericifolium. It
is
also “heather-leafed;” and its antecedents too are surmised to
have faced
the bitter chill at the ice’s edge some 300 to 250 million years ago.
Along with other changes in leaf morphology, the leaves of Banksiae
have evolved a whorl pattern, which is most in evidence on the densely
needled limbs of B. ericifolia. The whorl pattern very likely evolved
during
the ice age, because of a need to capture ice-melting sunlight more
effectively.
12
|
Figure 5: Banksia
spinulosa v cunningham mature flower spike

13
|
Figure 6: Banksia
integrifolia, showing increased leaf elaboration over Figures 1 and 5.

14
|
Figure 7: Banksia burdettii shows the most leaf elaboration of Figures
1,5,6 and 7,
presumably meaning this plant was further away from the ice fields than
others.

15
|
Furthermore, like many other
Proteaceae, the above Banksiae have hard, leathery leaves. However,
this hard-leaf (sclerophyllic) development
may have been less a response to the Gondwanaland Ice Age than an
adaptation later to semi-arid, sandy habitats, poor in mineral content
- - a
secondary evolutionary effect that followed the ice age in both
Australia and
Africa.
Leucospermum cordifolium, an
Afrop P-oide
Pointing now to the Afrops, consider first the genus Leucospermum,
among the best known of the Proteaceae. Leucospermum is commonly
called
a pincushion plant, and, indeed, it looks like a pincushion after its
long, stiff styles have flipped out. About 90% of all Leucosperma
grow today
among other woody shrubs on sandy hillsides and in fields termed
“fynbos”
within “the Cape Folded Mountain Region” of South Africa that
fronts the
Indian Ocean. The mature flower heads of Leucosperma are beautiful
(Figures 8 and 9). They range in color from chartreuse to deep orange,
from pink
to crimson. Figure 10 shows three newly budded flower heads of a
Leucospermum hybrid nestled among leathery leaves.
In Figure 11, an immature Leucospermum cordifolium flower head displays
long, stout styles with top-shaped pollen presenters. The flower
head clearly shows the stigmas atop the pollen presenters. Although
these stigmas
have taken on pollen, they may not yet be receptive to pollination.
In this immature L.cordifolium, the styles are flipping out, starting
at the periphery and advancing toward the center, in a gradual
sequential
manner (reminiscent of the gradual advance of the flipped out styles of
Banksia ericifolia in Figure 4). The less mature inner flowers are not
yet laid
out. Like B. ericifolia, they show a reentrant style. The pollen
presenter
at the far end of each “unflipped” style is surrounded by the four
anthers
attached within the perianth cup from which it will spring. A
tiny superior
ovary is present at the near end of the style. The anthers, styles,
stigmas,
pollen presenters, and ovaries resemble closely the sex organs of
B.ericifolia.
The similar, yet unique sex organs of the flowers of an Austrop G-oide
(B. ericifolia) and an Afrop P-oide (L. cordifolium) tell of a land
passage
long ago traveled by Proteaceae between Australia and Africa. This
remarkable sameness of floral elements has been preserved to the
present time
despite three hundred million years of separation and the vast
interposition of
the Indian Ocean.
16
|
Figure
8: Leucospermum, an
Afrop P-oide, is presented in Figures 8 through 13.
It grows
mainly in the sandy hills of the "Cape Folded Mountain Region" of South
Africa,
facing the Indian Ocean. There are 40 species. At top is
Leucospermum "Veldfire"
maturing flowerhead, at mag. x 1.7.

17
|
Figure 9: Leucospermum cordifolium maturing
flower head, at mag. x
1.2.

18
|
Figure 10: Leucospermum hybrid flower heads, newly sprung,
at mag. x 1.6.
The leaves are leathery.

19
|
Figure 11: L. cordifolium at several stages of maturity, at mag.
x
3.
Note the open stigmas atop the pollen presenters. At
periphery,
mature flowers have flipped out, central flowers are still
immature.

20
|
A Strikingly Disparate
Similarity
The sameness of Austrop and Afrop floral elements is wonderfully told
by two Proteaceae that are extremely different both in form and in
location. One is a ground-hugging Afrop shrub, Leucospermum gerrardii.
The other is a massive, towering Austrop tree, Grevillea robusta.
L. gerrardii is a leathery-leafed mat shrub found in the mountainous
regions of the eastern Transvaal, where the rainfall is plentiful in
summer.
Its floral tint changes from yellow (in Figure 12) to red (in Figures
13) in going
from youth to maturity. Grevillea robusta, on the other hand, is a
leafy
tree, an evergreen, mainly indigenous to Southwestern Australia. Termed
a silk
oak, it often grows to more than 20 meters in height and is about 1.6
meters
in girth. Several months of the year, this treeis conspicuously yellow
with flowers. The flowers grow only on the upper part of the
horizontally
inclined flower spike; and, characteristic of a G-oide, they can be
seen in
pairs in
Figure 14.
Most surprising is one detail. The flowers of both the Afrop shrub
(Figures 13) and the Austrop tree (Figure 15) display an inverted red-
colored underside of the perianth, like a four-striped red ribbon. In
both flowers, the perianth can be seen splitting lengthwise into four
characteristic segments, while the styles are in process of flipping
out. In
immaturity, the pollen presenters of the Afrop shrub and the Austrop
tree are sheathed
in their perianth cups (Figures 12 and 14, respectively). Best
seen
in Figure 12, the perianths of L. gerrardii are white-haired
– just as
are the
perianths of Austrop Banksia burdetti in Figure 7. The L. gerrardi
flower head is shown again at a later stage (Figure 13) where almost
all of
its conical pollen presenters are flipped out and covered with yellow
pollen.
These idiomorphic similarities of disparate, distant Australian and
African species bear important witness that the Proteaceae ranged
Gondwanaland from Australia to Indo-Madagascar to Africa to South
America prior to
the Gondwanaland Ice Age 300 million years ago. Grevillea robusta
displays a style (Figure 16) as it was just starting to hump its way
out of a
perianth while attached to it at both ends. A more mature flower
spike is
shown (Figure 17), just after its (deciduous) perianth segments had
been
blown away. The female sex organs were all there, bare and alone,
starkly
mounted on stems, offering a unique epitome of proteaceous parts: the
minute superior ovaries, the styles,
and the minute pollen presenters with stigmas – parts that
are common
to both Austrop and
Afrop.
21
|
Figure 12: Leucospermum gerrardii, an Afrop P-oide mat shrub,
showing immature flower head.
The styles are sheathed in
white-haired perianths (as are
the styles of Austrop G-oide
Banksia burdettii in Figure 7).

22
|
Figure 13: L. gerrardii, showing perianths, each with four inverted
("turned out") red striped segments. Observe that the pollen
presenters are coated with pollen.

23
|
Figure 14: Grevillea robusta, an Austrop G-oide tree, showing immature
flower spike at mag. x 5. Flowers are in pairs, on upper side of
spike only.

24
|
Figure
15: Grevillea robusta, immature flower, showing perianth
with four
"turned out" red-striped segments at mat. x 9. Compare with
Figure 13.

25
|
Figure 16: Grevillea robusta,
immature flower, with style just
starting to hump its way out of the
perianth, at mag. x 6.5.

26
|
Figure 17: G. robusta mature flower spike devoid of (deciduous)
perianths.
Each bare flower shows a unique
summary of its female parts:
the minute
(superior) ovary, the style, and
the pollen presenter with
stigma.
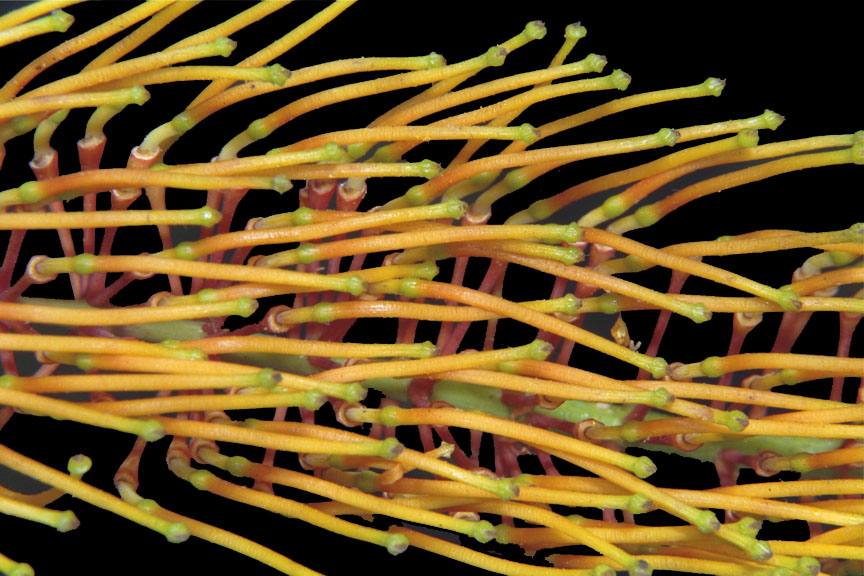
27
|
Ancestral Voices
Picture the Proteaceae during the Carboniferous already spread across
Gondwanaland, initially native to cool rainforests. There, the first
flowering plants had surely inherited fern-like leaves from fern
ancestors. At first, in continuity, they consociated with the seed
ferns and other
ferns, which
had reached their heyday. At that time the Gondwanaland flora from one
end
to the other was dominated by glossipterids (seed ferns that had
tongue-shaped fronds with large midribs). Even today at elevated
heights on the
volcanic soil of half-million-year-old Hawaii, ferns exert an awesome
influence. Hapu tree ferns and uluhe mat ferns dominate. With their
enormous
light- excluding fronds, they blanket the impenetrable forest floor
with
darkness, as their ancestors likely did during the Carboniferous. It
seems
plausible that the Proteaceae gradually spread out, or were crowded out
by the ferns,
into less hospitable – open and semi-arid –
environments. The
towering
G. robusta trees were probably among the last to depart the cooling
rainforests.
Morphological Retrenchment
To varying degrees, ancestral fern characteristics are conserved among
the Proteaceae. Ronald Melville reported incipient transitions from
seed
ferns to angiosperms in 1960 (2) and in later papers. “The primary
diversification of the pre-angiosperm stock appears to have taken
place,” he inferred, “ . . . during the late Carboniferous or early
Permian. Many angiosperm lineages must date back to this period as
distinct lines of
evolution . . .” He noted that the parallel
veining found in the leaves of Proteaceae
and in other primitive plants could have evolved from Gangamopteris, an
extremely ancient (Devonian) Gondwanaland seed fern (without a large
frond midrib).
The leaves of G. robusta in Figures 18 and 19 are
offset-opposite, long, broad, comparatively soft, thin, and deeply
divided (in contrast to
the whorled needles of Banksia ericifolia in Figure 1). Species
like G.
robusta show, “primitive morphological characters” when they have large
compound leaves with expanded leaflets, according to Venkata Rao, 1971
(3).
Large, compound leaves indicate a mainly frost-free past.
28
|
The leaves of G. robusta show a
fern-like pattern and a trace of parallel veining; and, indeed, these
leaves appear to resemble the leaf patterns
of the spleenworts, the woodsias, the spear-leaved fern, and other
ferns alive
today. Furthermore, parallel veining is manifest in the leaves of
Banksia
burdettii (Figure 7). And here too appears a resemblance to the leaves
of the
fern genus Pteris, with their slender shape and large frond midrib.
In premise, the Permo-Carboniferous Ice Age eroded much of the
ancestral parallel veining, starting with the approach of the ice 300
million
years ago. To argue the deletion of parallel veining, a leaf form
sequence for
Banksiae is cited once again, starting with the parallel veining in
evidence in
Banksia burdettii and following in sequence with the leaf of Banksia
speciosa, now introduced as Figure 20, to represent an early incursion
into
parallel
veining. B.speciosa is a large shrub, endemic to Australian
near-coastal sand
dunes.
Examining a magnified undersurface of its leaf, the parallel veining is
shown starting outward at right angles to the midrib (as in B.
burdettii),
but then, converging to a point in each segment. Moreover, with the
intrusion of
the ice age, this leaf lost its serrations (so evident in B.
burdettii);
and it became deeply divided and smooth. Farther into icy regions, the
leaf of B. integrifolia is smooth and linear (Figure 6). And in the
shadow of
glaciers in New South Wales, then well within the Antarctic Circle,
B.ericifolia
(Figure 1) had evolved needles.
And Banksiae are not alone in transition from large leaves to needles
during the Permo-Carboniferous Ice Age. Grevilleae too, in Australia,
simplified their leaves. Indeed, among the 273 species of Grevilleae,
leaf form sequences are readily observable that denote simplification.
For
example, far away from the ice were the long, broad leaves of G.
robusta in Figure
18. Closer in were smaller leaves represented by the hybrid Grevillea
“Robyn Gordon,” (Figure 21), which bear resemblance to those leaves of
G.
robusta. And ever closer were the deeply parted leaves of Grevillea
boongala (or spinebill), in Figure 22. They resemble the segmented
leaves of B.
speciosa in Figure 20. In apparent remarkable convergence, the hybrid
Grevillea
“Red Glow” (Figure 23) shares the needle badge of nearness with Banksia
ericifolia (Figure 1) – both having faced up to the
Gondwanaland ice
and survived.
In summary, the leaves of present-day Proteaceae, within the two genera
examined, display a sequence of evolutionary curtailment of form and
structure apparently in adaptation to gradual but varied encroachments
of icefields. Before the
Permo-Carboniferous Ice Age, large compound leaves with
(seed-fern-remanent) parallel veining were characteristic of
Proteaceae. However, during that ice age, leaf simplification occurred
within
genera, from large compound leaves to needles (at the extreme).
29
|
Figure 18: G. robusta leaf pattern, suggestive of fern frond, mag
x
0.7

30
|
Figure
19: G. robusta with indications of parallel venation, mag x. 1.3

31
|
Figure 20: Banksia speciosa, underside of leaf, mag. x 4.7, at right.
The
veining
starts as parallel, but comes to a point. Left photo shows
segmented leaves at different ages.

32
|
Figure
21: Hybrid shrub Grevillea "Robyn Gordon" leaf at mag. 1.7.
Note
resemblance to G. robusta leaf in Figure 19.

33
|
Fig. 22: Grevillea boongala (or spinebill). Deeply parted leaves
show
inroads of glaciation. Compare to segmented leaves of B. speciosa,
Figure 20.

34
|
Figure 23: Grevillea "Red Glow" needles, mag. x. 2,
indicating extreme
adaptation to glaciation.

35
|
Yet even today, some species of Proteaceae that were less
exposed to
ancient cold, have large leaves with deeply divided or expanded
leaflets that are, as
Venkata Rao noted (3), primitive characters of Proteaceae. Moreover,
evidence
of parallel veining, associated with seed fern ancestry, remains in
species with the most frost-free pasts.
The Nectar Factor
The perianth of most Afrops and Austrops produces copious nectar that
lures insects, birds, and mammals to pollinate flowers. The perianth is
both
the nectar source and its reservoir. On the inside of its tube wall,
below
the enclosed tiny ovary, are four scale-like glands (collectively a
nectary), which fill the inside base of the perianth with nectar. The
perianth’s nectar
reservoir is in slight view in the second frontispiece.
Nectar reservoirs are in the flowers of most Afrop and Austrop shrubs
and trees and are essential to their reproduction. Over the past 360
million years, they undoubtedly have had a key and even crucial role in
the
co-evolution of a wide variety of pollination-capable insects, as well
as other fauna.
Insect and angiosperm early adapted to each other’s needs for mutual
benefit.
The survival of the Proteaceae tothis present day is due largely to the
beneficial exchanges of pollination and reward. The impact of these
exchanges on faunal evolution is discussed in companion
essays.
The Flower That Blooms This Day
Like the mythical god Proteus, the Proteaceae come in a wide variety of
shapes and sizes; yet they present unique constancy of reproductive
organs identical in essential form and function. Their flower heads
range from
gaunt to lovely, and the latter quality can be savored in Grevillea
banksii
(Figures 24 and 25) and Grevillea longistyla (Figure 28). An
elaborately
convoluted perianth is displayed by G. longistyla in Figure 29. Both G.
banksii
(Figure 26) and G. longistyla (Figure 30) clearly reveal the uniquely
proteaceous anther-to-perianth cup attachment. Immature G. banksii
fruit follicles
(with one or two seeds) are shown in both Figure 24 and 27. The ornate
flowers of G. longistyla tell that its antecedents had eluded the
intense chill of
the Permo-Carboniferous Ice Age. Contrast once again the gaunt Banksia
ericifolia flowers in Figure 2.
36
|
Figure 24: This is Grevillea banksii "Forsters," flower spike
(and
in the background, immature fruit follicles).

37
|
Figure 25: Grevillea banksii flowers in various stages of maturity,
mag.
x 3

38
|
Figure 26: Grevillea banksii perianth cup after release of style
at mag.
x 7. Note the anthers within the cup.

39
|
Figure 27: Immature fruit and follicles of Grevillea banksii.

40
|
Figure
28: Grevillea longistyla, ornately convoluted perianths, mag. x 3.
Compare with simplified perianths of Banksia ericifolia in Figures 2
and 3.

41
|
Figure 29: G. longistyla flowers show bilateral symmetry of perianth,
at
mag. x 5.
On each side of emerging style,
an inner perinth segment is
nested within an outer one.

42
|
Figure 30: G. longistyla, with
"curled back" perianth after style release.
It shows three of its four
anthers adnate to perianth segments at mag. x 5.5.

43
|
Summary
By reason of the angiosperm invention of the bisexual flower, the
Proteacea found a stable independence, an enhanced ability to
diversify, and a
greater freedom to roam far and wide. The present worldwide
distribution of Proteaceae (shown in third frontispiece) is sometimes
termed
“enigmatic” and is, indeed, wondrous. It is a remarkable end product of
the Permo- Carboniferous Ice Age and the breakaway of a large landmass
from Australia, coupled with the gradual drifting apart of the
component Gondwanaland continents. The unique reproductive organ
hallmark of the Proteaceae and their enigmatic world-wide distribution
strongly affirm
that the Proteaceae inhabited the entire length of Gondwanaland, from
Australia to South America, more than 300 million years ago.
References
1) Vogts, M., Proteaceae, C. Struik, Cape Town, South Africa, 1982.
2) Melville, R., Glossipteridae, Angiospermidae, and the evidence
of
angiosperm’s origin, Bull. J. Linn. Soc., 86,
279-323 (1983).
3) Rao, C.V., Botanical Monograph No. 6 Proteaceae, Council of
Scientific
and Industrial Research, New Delhi, 1971.
4) Tarling, D.H. and Tarling, M.P., Continental Drift, a Study of the
Earth’s
Moving Surface, G. Bell and Sons, London,
1971.
5) Metcalfe, I., “Origin and assembly of South-east Asian
continental
terranes,” 101-118, in Gondwana and Tethys,
eds. M.G. Audley-
Charles and A. Hallam, Geological Society,
Oxford Press, 1988.
44
|


































